The Hidden Reason Cancer Drugs Fail: Lysosomal Reservoirs and the Future of Personalized Treatment
For years, oncologists have grappled with a frustrating reality: the same cancer treatment can yield dramatically different results for different patients. Now, groundbreaking research led by Dr. Louise Fets at the MRC Laboratory of Medical Sciences (LMS) is shedding light on a key factor – the unexpected role of lysosomes, cellular “recycling centers,” in trapping and releasing cancer drugs.
Uneven Drug Distribution: A Major Obstacle
The study, published in Nature Communications, focuses on PARP inhibitors, a class of targeted therapies that have revolutionized ovarian cancer treatment. Researchers discovered that these drugs don’t always reach their intended targets within cancer cells. Instead, they can accumulate inside lysosomes, creating internal reservoirs that lead to uneven drug distribution. Some cells receive a potent dose, while others are barely affected.
This variability isn’t just a theoretical concern. It directly impacts treatment efficacy and the development of drug resistance. “We were surprised to see large variability in drug accumulation at the single-cell level,” explains Dr. Carmen Ramirez Moncayo, a postdoctoral researcher at the LMS. “This variability was driven by the build-up of a drug in lysosomes, which are acting as reservoirs.”
How Lysosomes Impact Drug Effectiveness
Lysosomes, traditionally understood for their role in cellular waste disposal, are now recognized as dynamic players in drug metabolism. Certain PARP inhibitors, like rucaparib and niraparib, are particularly susceptible to being sequestered within these compartments. This creates a slow-release mechanism, altering the drug’s availability and potentially diminishing its impact.
Interestingly, not all PARP inhibitors are affected equally. Olaparib, for example, appears to bypass this lysosomal trapping mechanism. This suggests that the chemical properties of the drug itself play a crucial role in determining its fate within the cell.
Mapping Drug Delivery with Advanced Imaging
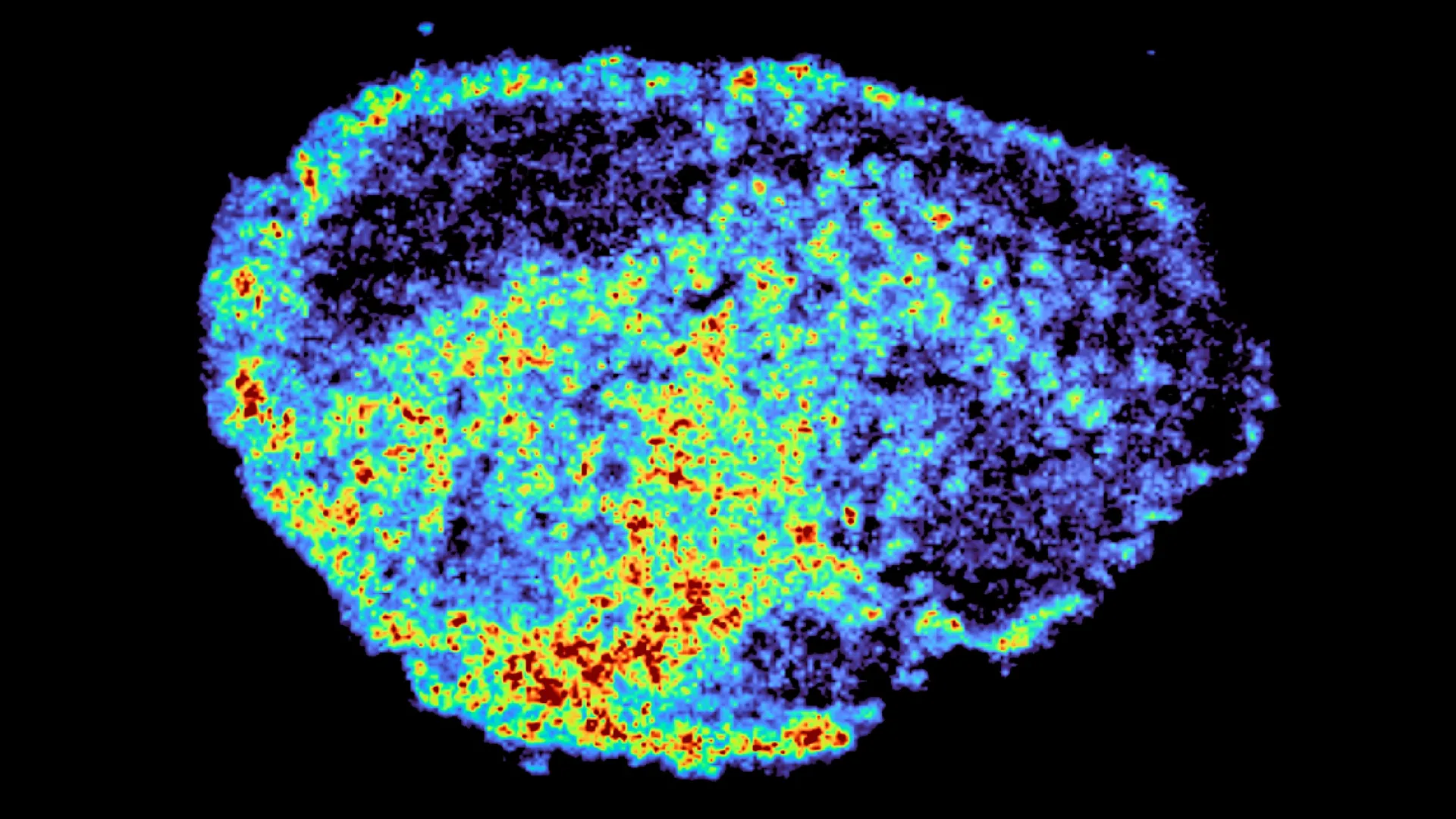
The research team employed cutting-edge techniques, including mass spectrometry imaging and spatial transcriptomics, to visualize drug distribution within patient-derived ovarian tumor samples. These methods allowed them to create detailed maps showing exactly where drug molecules accumulated and to correlate drug levels with gene activity.
“A novel aspect of this study was the use of mass spectrometry imaging to directly measure and visualize drug uptake in patient tumour tissue,” says Dr. Zoe Hall, from Imperial’s Department of Metabolism, Digestion and Reproduction. “Through the spatial mapping of drug molecules, we could pinpoint regions of high and low drug and compare gene expression, from the same tissue slice, using spatial transcriptomics.”
Future Trends: Towards Personalized Cancer Therapy
This discovery opens up exciting possibilities for personalized cancer treatment. By understanding how drugs are processed within individual tumors, doctors may be able to tailor treatment strategies to maximize effectiveness and minimize resistance.
Here are some potential future trends:
- Biomarker Identification: Identifying biomarkers that predict which patients are most likely to benefit from specific PARP inhibitors, based on their lysosomal activity and drug metabolism profiles.
- Drug Repurposing: Exploring the potential of combining PARP inhibitors with drugs that modulate lysosomal function, either to enhance drug delivery or to overcome resistance.
- Novel Drug Design: Developing new PARP inhibitors that are less susceptible to lysosomal sequestration, ensuring more consistent drug exposure within cancer cells.
- Advanced Imaging for Treatment Monitoring: Utilizing advanced imaging techniques to monitor drug distribution in real-time during treatment, allowing for adjustments to dosage or therapy selection.
Dr. Louise Fets envisions a future where molecular signatures of a patient’s tumor are used to guide therapeutic decisions. “By understanding how drugs are taken up into cells, we can understand whether this influences why cancer drugs work for some people and not for others. Eventually, we hope to be able study the molecular signature of a patient’s tumor to help to tailor therapeutic approaches in a more personalized way.”
Beyond Ovarian Cancer: Implications for Other Cancers
While this research focused on ovarian cancer, the principles of lysosomal drug sequestration likely apply to other cancer types as well. PARP inhibitors are also used to treat breast and prostate cancers and are being investigated for use in many others. Understanding these fundamental mechanisms of drug distribution could have broad implications for cancer treatment across the board.
Frequently Asked Questions
Q: What are lysosomes?
A: Lysosomes are cellular structures that act as “recycling centers,” breaking down waste materials and cellular debris.
Q: What are PARP inhibitors?
A: PARP inhibitors are a class of targeted cancer drugs that block an enzyme involved in DNA repair, making cancer cells more vulnerable to treatment.
Q: How does this research impact patients?
A: This research could lead to more personalized cancer treatments, ensuring that patients receive the most effective therapies based on their individual tumor characteristics.
Q: Is this research still in early stages?
A: While promising, this research is ongoing. Future studies will involve animal models and larger patient groups to validate these findings and translate them into clinical practice.
Did you know? The uneven distribution of cancer drugs within tumors can be visualized using advanced imaging techniques like mass spectrometry, revealing “hotspots” of high drug concentration and “coldspots” with minimal drug exposure.
Pro Tip: Discuss your treatment options and potential side effects with your oncologist. Understanding the nuances of your specific cancer and the drugs being used is crucial for informed decision-making.
Stay informed about the latest advancements in cancer research. Explore more articles on our website and subscribe to our newsletter for updates.