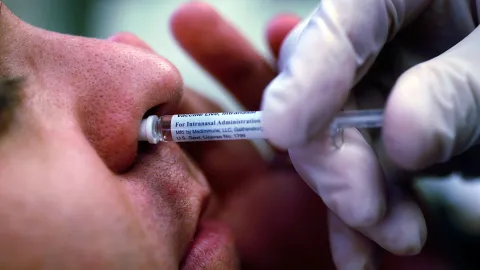
The Future of Respiratory Protection: Stanford’s Needle-Free Nasal Spray Vaccine
A groundbreaking nasal spray vaccine developed by Stanford University scientists promises a paradigm shift in how we approach preventing respiratory illnesses and allergies. Published in Science in 2026, the research details a single-application spray capable of providing broad protection, potentially transforming future immunization campaigns.
Beyond Traditional Vaccines: A Multi-Pronged Approach
Unlike conventional vaccines that target specific pathogens, this innovative nasal spray stimulates multiple defense mechanisms simultaneously. It activates the innate immune system for immediate response although likewise bolstering immunological memory for long-term protection. This approach mimics a controlled natural infection, keeping the lungs primed to defend against viruses, bacteria, and allergens.
In animal trials with mice, a single application reduced viral load in the lungs by up to 700 times. Importantly, the mice did not exhibit severe side effects like significant inflammation or weight loss, suggesting a promising early safety profile.
Broad-Spectrum Protection: More Than Just Viruses
The vaccine’s potential extends beyond viral infections. Research demonstrated protection against bacterial infections and a reduction in allergic reactions to common allergens. This broad-spectrum capability could significantly simplify preventative healthcare.
The formula combines components that activate different parts of the immune system, including innate response stimulators, proteins attracting T cells to the lungs, and signals prolonging protection. This combination trains the body to react to multiple respiratory threats, rather than focusing on a single target.
Timeline to Implementation: From Lab to Public Health
Currently in the pre-clinical phase, the nasal spray vaccine has not yet been tested on humans. Researchers anticipate clinical trials could begin between late 2026 and 2027 to assess safety and efficacy. Positive results would pave the way for large-scale studies between 2028 and 2029, with potential approval within the following five to seven years.
Logistical Advantages: Simplifying Immunization
Administering a vaccine via nasal spray could dramatically simplify logistics, particularly for populations with a fear of needles or limited access to healthcare professionals. Eliminating the need for syringes and complex application procedures could increase coverage and reduce costs for public health systems.
The Stanford Drug Discovery Symposium and Blockchain’s Role in Pharma
Stanford University continues to be a hub for biomedical innovation. The 10th annual Stanford Drug Discovery Symposium (SDDS 2026), held at the Arrillaga Alumni Center, brings together researchers, pharmaceutical companies, and investors. Interestingly, the Science of Blockchain Conference 2026 will also be held at Stanford University in July, suggesting a growing interest in the intersection of blockchain technology and pharmaceutical advancements.
Blockchain and the Future of Drug Supply Chains
While not directly related to the nasal spray vaccine, the concurrent blockchain conference highlights a potential future trend: increased use of blockchain technology to enhance drug supply chain security and transparency. This could help combat counterfeit drugs and ensure the integrity of the pharmaceutical distribution process.
Advances in Cartilage Regeneration: A Parallel Breakthrough
Alongside advancements in vaccine technology, Stanford Medicine researchers have also made significant strides in cartilage regeneration, potentially offering a solution for arthritis. This discovery, published in January 2026, involves blocking a protein linked to aging to restore healthy cartilage in aging joints and prevent arthritis after knee injuries.
The Convergence of Immunological and Regenerative Medicine
These parallel breakthroughs in immunology and regenerative medicine demonstrate Stanford’s commitment to tackling major health challenges through innovative research. The convergence of these fields could lead to even more effective and comprehensive treatments in the future.
FAQ
Q: When will this nasal spray vaccine be available to the public?
A: Clinical trials are expected to begin between late 2026 and 2027. If successful, it could be available within five to seven years.
Q: Is this vaccine safe?
A: Early animal trials show a promising safety profile with no severe side effects observed.
Q: Does this vaccine protect against all respiratory illnesses?
A: The research suggests broad-spectrum protection against viruses, bacteria, and allergens, but further testing is needed to confirm its effectiveness against all respiratory threats.
Q: Where can I find more information about the study?
A: Details of the study and its publication are available on the Science journal website: https://www.science.org
Did you know? Stanford University is also hosting the Science of Blockchain Conference 2026, indicating a growing focus on the intersection of technology and healthcare.
Pro Tip: Staying informed about emerging medical technologies is crucial for proactive healthcare. Regularly consult reputable sources like ScienceDaily and peer-reviewed journals.
Interested in learning more about cutting-edge medical research? Explore our other articles on biomedical innovation and public health. Share your thoughts in the comments below!