Blood Test Breakthrough: Predicting Abnormal Bone Growth Before It Happens
Dallas, TX – A new blood test developed by researchers at UT Southwestern Medical Center promises to revolutionize the detection of heterotopic ossification (HO), the abnormal formation of bone in soft tissues. Published in Nature Communications on March 24, 2026, this test can identify HO weeks before traditional imaging methods, potentially leading to earlier treatment and improved patient outcomes.
Understanding Heterotopic Ossification
HO is a common complication following trauma or certain surgeries, like hip replacements. The body’s natural healing process goes awry, resulting in bone growth in muscles, tendons, and other soft tissues. Symptoms range from pain and swelling to joint stiffness and reduced range of motion. Currently, diagnosis relies on detecting bone formation on CT scans or X-rays, a process that can seize six to eight weeks.
The Power of Liquid Biopsy: A New Approach
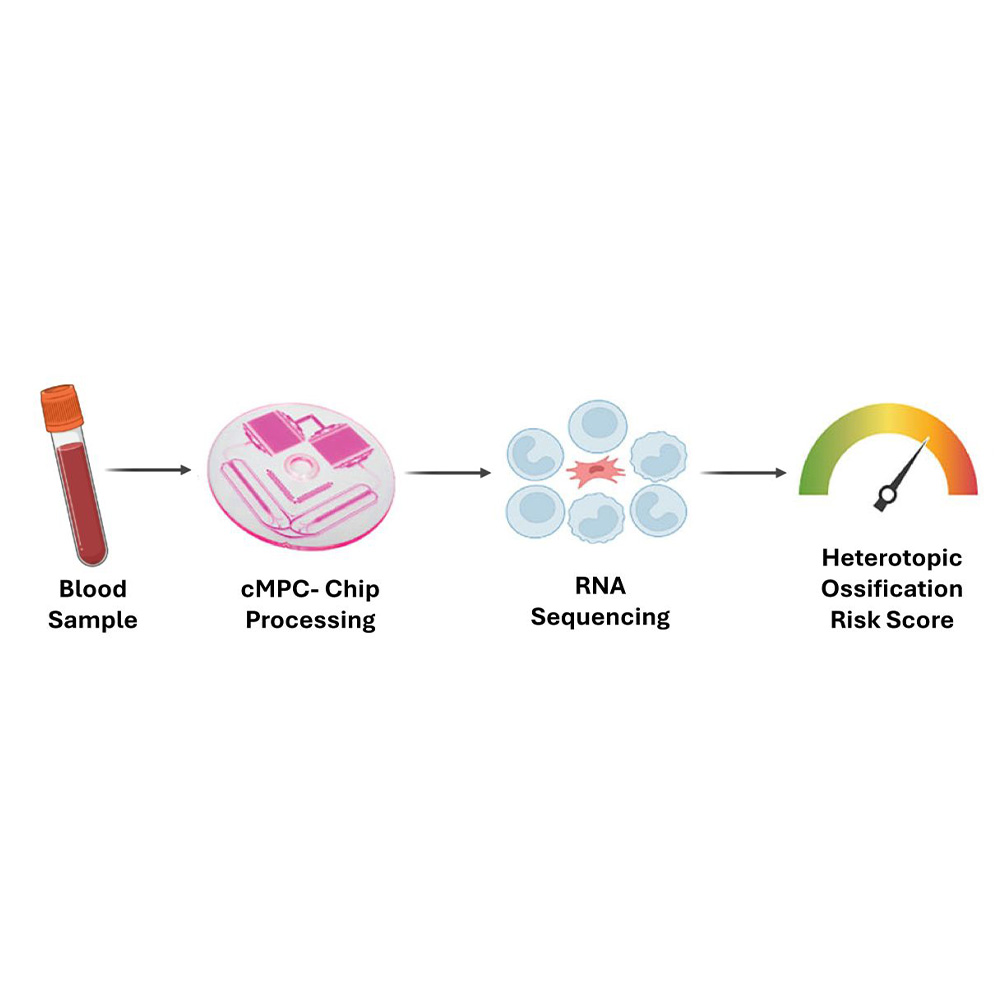
The UT Southwestern team, led by Dr. Benjamin Levi and Dr. Johanna Nunez, focused on circulating mesenchymal progenitor cells (cMPCs) – rare cells in the blood involved in abnormal bone formation. Using a microfluidic device called an iChip, researchers isolated these cMPCs from both mouse models and patients who had undergone hip replacement surgery. Analyzing the cells’ gene expression allowed them to build a risk prediction model with 90% accuracy.
Pro Tip: This “liquid biopsy” approach offers a less invasive and faster alternative to traditional imaging, potentially changing how HO is managed.
Reducing Unnecessary Treatment
Currently, a significant number of patients receive preventative treatments – such as radiation or nonsteroidal anti-inflammatory drugs – even if they don’t develop HO. This represents due to the delay in diagnosis. Dr. Nunez explains that this new blood test could reduce unnecessary interventions, lowering costs, complications, and associated risks. “A blood-based ‘liquid biopsy’ tool like this could allow early identification, reducing complications, costs, overtreatment, and associated risks while improving precision, monitoring, and safety,” she stated.
Monitoring Treatment Response
Beyond early detection, the test can also be used to monitor how patients respond to treatment. By tracking changes in cMPC gene expression, clinicians can assess whether interventions like physical therapy, radiation, or medication are effectively reducing the risk of HO.
Future Trends in Early Bone Growth Detection
The development of this blood test signals a broader trend toward proactive and personalized medicine. Here’s what we can expect to see in the coming years:
- Expansion to Other Conditions: The principles behind this test – identifying specific cellular markers in the blood – could be applied to detect other types of abnormal bone growth or even early stages of bone diseases like osteoarthritis.
- AI-Powered Diagnostics: Artificial intelligence and machine learning will play an increasingly important role in analyzing complex gene expression data, improving the accuracy and speed of diagnosis.
- Point-of-Care Testing: The goal is to develop portable, easy-to-use versions of this test that can be administered in clinics or even at the bedside, providing rapid results.
- Personalized Prevention Strategies: Identifying individuals at high risk of HO will allow for tailored prevention strategies, minimizing the need for broad-spectrum treatments.
Did you know?
Up to 80% of patients who don’t develop HO may receive unnecessary preventative treatment after trauma or surgery.
Frequently Asked Questions
- What is heterotopic ossification? It’s the abnormal formation of bone in soft tissues, often after injury or surgery.
- How accurate is the new blood test? The test demonstrated 90% accuracy in identifying HO formation in preclinical models and patient samples.
- When will this test be widely available? The test is currently undergoing further research to evaluate its effectiveness and will require regulatory approval before it can be widely implemented.
- Is this test covered by insurance? Insurance coverage will depend on regulatory approval and individual insurance plans.
This research builds upon previous work by the Levi Lab and has resulted in a patent-pending diagnostic test. Further research is underway to refine and validate the test for broader clinical use.
Want to learn more about bone health and regenerative medicine? Explore additional articles on our website or subscribe to our newsletter for the latest updates.