Beyond the Pandemic: The New Frontier of mRNA Medicine
The global success of COVID-19 vaccines was merely the opening act. While mRNA technology made household names of companies like Moderna and Pfizer-BioNTech, the true revolution is only just beginning. We are moving from a world where mRNA is synonymous with “vaccine” to one where it functions as a versatile, programmable software for the human body.
By leveraging the body’s own cellular machinery to produce therapeutic proteins, researchers are unlocking treatments for conditions that were previously considered “undruggable.” From rare metabolic disorders to personalized cancer therapies, the next decade of biotechnology will be defined by how we refine, deliver, and design these genetic blueprints.
Precision Engineering: The Art of mRNA Design
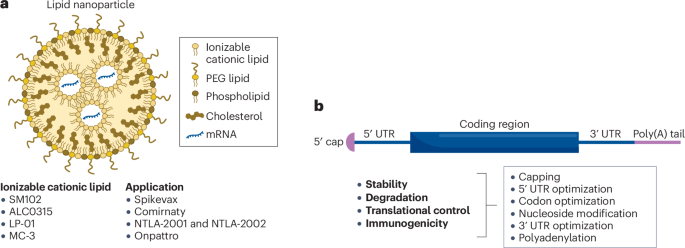
Modern mRNA therapeutics are not just simple sequences; they are highly engineered constructs. Scientists are now using deep learning algorithms to optimize every component of the mRNA molecule, from the 5′ cap and untranslated regions (UTRs) to the codon sequence itself.
By optimizing these elements, developers can increase the stability and translational efficiency of the mRNA, ensuring that the body produces the right amount of protein at the right time. Recent advancements in CleanCap® technology and nucleoside modifications, such as N1-methylpseudouridine, have already proven vital in reducing unwanted immune responses while maximizing protein yield.
Personalized Cancer Vaccines: Mobilizing the Immune System
Perhaps the most exciting application of mRNA lies in oncology. Rather than a “one-size-fits-all” approach, we are seeing the rise of individualized neoantigen therapies. By sequencing a patient’s specific tumor and identifying unique mutations, doctors can create a bespoke mRNA vaccine that trains the immune system to hunt down cancer cells with surgical precision.
In trials for melanoma and pancreatic cancer, these personalized vaccines have shown the ability to prime long-lived CD8+ T cells. This isn’t just treating the disease; it is effectively teaching the body to maintain its own surveillance system, potentially preventing recurrences that have plagued cancer survivors for decades.
Solving the Delivery Puzzle
The “Achilles’ heel” of mRNA has always been delivery. How do you get a fragile molecule into a specific cell without it being destroyed by the body’s natural defenses? The answer lies in next-generation lipid nanoparticles (LNPs).
Researchers are currently developing “organ-specific” LNPs. By tweaking the chemical structure of ionizable lipids, scientists can now direct mRNA to the liver, the lungs, or even the bone marrow. This precision reduces off-target side effects and opens the door for treating systemic diseases like glycogen storage disease or even cardiovascular conditions.
Gene Editing: The Ultimate Upgrade
The marriage of mRNA and CRISPR-Cas9 technology is changing the landscape of genetic medicine. Instead of using viral vectors—which can trigger immune reactions—scientists are using mRNA to deliver the “instructions” for gene-editing tools. This transient expression is safer and more controlled, as the editing machinery disappears once the job is done.
We are already seeing the first generation of in vivo base editing trials targeting high cholesterol and rare liver conditions. This represents the shift toward “N-of-1” medicine, where therapies can be tailored to the specific genetic makeup of an individual patient.
Frequently Asked Questions (FAQ)
Q: Are mRNA vaccines safe for long-term use?
A: mRNA is naturally degraded by the body shortly after the protein is produced. It does not integrate into your DNA, and the technology has been refined over two decades to minimize inflammatory responses.
Q: What diseases can mRNA technology treat besides COVID-19?
A: Clinical trials are currently underway for influenza, RSV, CMV, various cancers, cardiovascular diseases, and rare metabolic conditions like methylmalonic acidemia and glycogen storage disease.
Q: How do personalized cancer vaccines work?
A: These vaccines are designed by analyzing the genetic mutations in a patient’s tumor. The mRNA instructs the patient’s cells to produce proteins specific to those mutations, “teaching” the immune system to recognize and attack the cancer.
Q: What is the biggest challenge facing mRNA medicine today?
A: The primary challenge remains the delivery mechanism. Improving the stability of lipid nanoparticles and ensuring they reach the correct tissues without inducing toxicity is the current focus of intense global research.
The mRNA revolution is moving rapid. If you want to stay ahead of the curve on how these genetic therapies are reshaping modern medicine, subscribe to our weekly newsletter for exclusive updates on clinical trial breakthroughs and biotech industry trends. Have a question about a specific mRNA application? Leave a comment below!