Gotistobart: A Potential Turning Point for Advanced Squamous Lung Cancer?
For patients battling metastatic squamous non-small cell lung cancer (sqNSCLC) who have exhausted other treatment options, a new horizon may be emerging. Early results from the PRESERVE-003 trial, published in Nature Medicine, suggest that gotistobart, a novel anti-CTLA-4 antibody, could significantly improve survival rates compared to standard chemotherapy with docetaxel.
Understanding the Challenge: Immunotherapy Resistance
Lung cancer remains the leading cause of cancer death worldwide. While immunotherapy, specifically PD-1/PD-L1 inhibitors, has revolutionized treatment for many, a substantial portion of patients don’t respond initially, or develop resistance after a period of benefit. This is particularly true for those with sqNSCLC who have progressed after both immunotherapy and platinum-based chemotherapy – a group facing a particularly grim prognosis.
How Gotistobart Works: Targeting the Tumor Microenvironment
Gotistobart takes a different approach. Unlike traditional CTLA-4 inhibitors, it’s designed to selectively deplete regulatory T cells (Tregs) within the tumor microenvironment. Tregs are known to suppress the immune response, effectively shielding cancer cells from attack. By removing this shield, gotistobart aims to unleash the power of the immune system to fight the cancer. It’s a pH-sensitive antibody, meaning its activity is enhanced in the acidic environment of tumors.
PRESERVE-003: Stage 1 Results – A Promising Sign
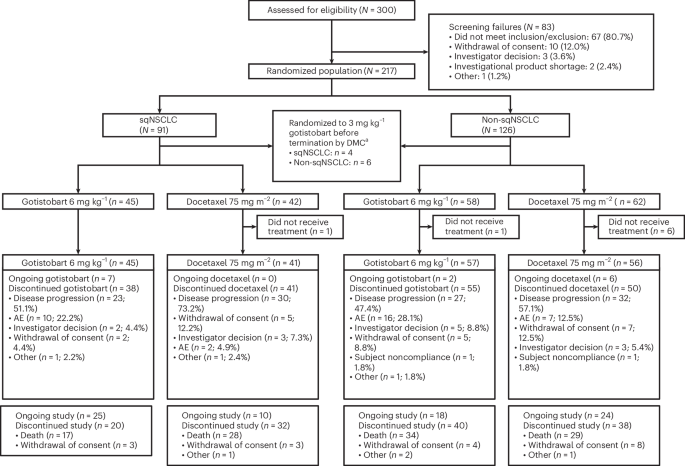
The PRESERVE-003 trial is a phase 3 study designed to evaluate gotistobart’s efficacy and safety. Stage 1 of the trial, involving 87 patients with squamous histology, showed a hazard ratio of 0.46 for death, meaning patients treated with gotistobart had a 54% lower risk of death compared to those receiving docetaxel. Median overall survival was not yet reached in the gotistobart arm, while it was 10.0 months with docetaxel. These results, while preliminary, are highly encouraging.
Importantly, the safety profile of gotistobart appeared manageable, with grade 3 or higher treatment-related adverse events occurring in 42% of patients receiving gotistobart versus 49% receiving docetaxel.
Beyond Survival: Other Potential Benefits
While overall survival is the primary endpoint, researchers are also evaluating progression-free survival, objective response rate, and duration of response. These secondary endpoints will provide a more comprehensive understanding of gotistobart’s impact on the disease.
Did you know? Regulatory T cells (Tregs) can make up a significant proportion of the cells within a tumor, actively suppressing the immune system’s ability to recognize and destroy cancer cells.
Future Trends and the Evolution of Lung Cancer Treatment
The PRESERVE-003 trial highlights a growing trend in cancer research: moving beyond broad immune activation to more targeted approaches. The focus is shifting towards modulating the tumor microenvironment to enhance the effectiveness of immunotherapy. This includes strategies to deplete immunosuppressive cells like Tregs, as well as approaches to increase the infiltration of immune cells into the tumor.
Combination therapies are also likely to play a crucial role. Researchers are exploring whether combining gotistobart with other immunotherapies, or even with targeted therapies, could further improve outcomes. The development of biomarkers to predict which patients are most likely to benefit from gotistobart will also be essential.
FAQ
Q: What is sqNSCLC?
A: Squamous non-small cell lung cancer is a subtype of lung cancer characterized by specific cellular features.
Q: What does “not reached” mean for median overall survival?
A: It means that, at the time of analysis, half of the patients in that group were still alive, and the median survival time hasn’t been determined yet.
Q: Is gotistobart a cure for lung cancer?
A: While the results are promising, it’s too early to say if gotistobart is a cure. Further research is needed to confirm these findings and determine the long-term benefits.
Q: What is a CTLA-4 inhibitor?
A: CTLA-4 inhibitors are a type of immunotherapy that blocks the CTLA-4 protein, which can help the immune system attack cancer cells.
Pro Tip: Staying informed about the latest clinical trials and treatment options is crucial for patients with advanced cancer. Discuss your options with your oncologist.
The PRESERVE-003 trial represents a significant step forward in the fight against advanced sqNSCLC. As the trial progresses and more data become available, gotistobart could potentially offer a much-needed new treatment option for patients who have exhausted other possibilities.
Aim for to learn more? Explore other articles on immunotherapy and lung cancer treatment on our website. Share your thoughts and questions in the comments below!