The End of the Daily Pill? How RNAi is Redefining Hypertension Treatment
For decades, managing high blood pressure has been a test of endurance. It is a daily ritual of pills and reminders, where success depends entirely on a patient’s memory and discipline. Yet, despite the availability of effective drugs, the global success rate is surprisingly low.

Pooled global analyses from 1990 to 2019 reveal a sobering reality: in 2019, fewer than 25% of people with hypertension actually achieved controlled blood pressure levels. The problem isn’t a lack of medicine; it’s the “adherence trap.”
As hypertension is often asymptomatic—meaning you can’t “perceive” your blood pressure rising—there is no immediate physiological reward for taking a pill. This creates a system where cardiovascular protection becomes a social filter, tracking a patient’s life stability rather than their actual clinical need.
Enter Zilebesiran: The “Vaccine-Like” Shift in Care
We are now seeing the emergence of a paradigm shift. Modern long-acting RNA interference (RNAi) therapies, such as zilebesiran, are moving us away from daily behavioral achievements and toward scheduled, system-mediated protection.
Zilebesiran works by targeting hepatic angiotensinogen (AGT), suppressing a critical upstream rate-limiting step in the renin-angiotensin-aldosterone system (RAAS). In simpler terms, instead of blocking the system every day, this therapy “silences” the production of a key protein that drives blood pressure up.
The result? A single subcutaneous dose can sustain lower blood pressure levels for several months. This transforms the responsibility of care from the patient’s memory to the healthcare system’s reliability.
Breaking Down the Clinical Evidence
The potential of this technology is being mapped out through several key clinical trials. The KARDIA-1 phase 2 trial demonstrated that dosing every three or six months could lead to persistent reductions in systolic blood pressure.

However, the road to innovation is rarely a straight line. In the KARDIA-3 trial, which focused on higher-risk patients, the primary endpoint—placebo-adjusted office systolic blood pressure lowering at month three—did not meet statistical significance after multiplicity adjustment.
The next major milestone is ZENITH, an upcoming global phase 3, event-driven trial. Expected to enroll approximately 11,000 patients, ZENITH will determine if twice-yearly angiotensinogen silencing can actually reduce major events, including cardiovascular death, nonfatal stroke, nonfatal myocardial infarction, and heart failure when added to standard care.
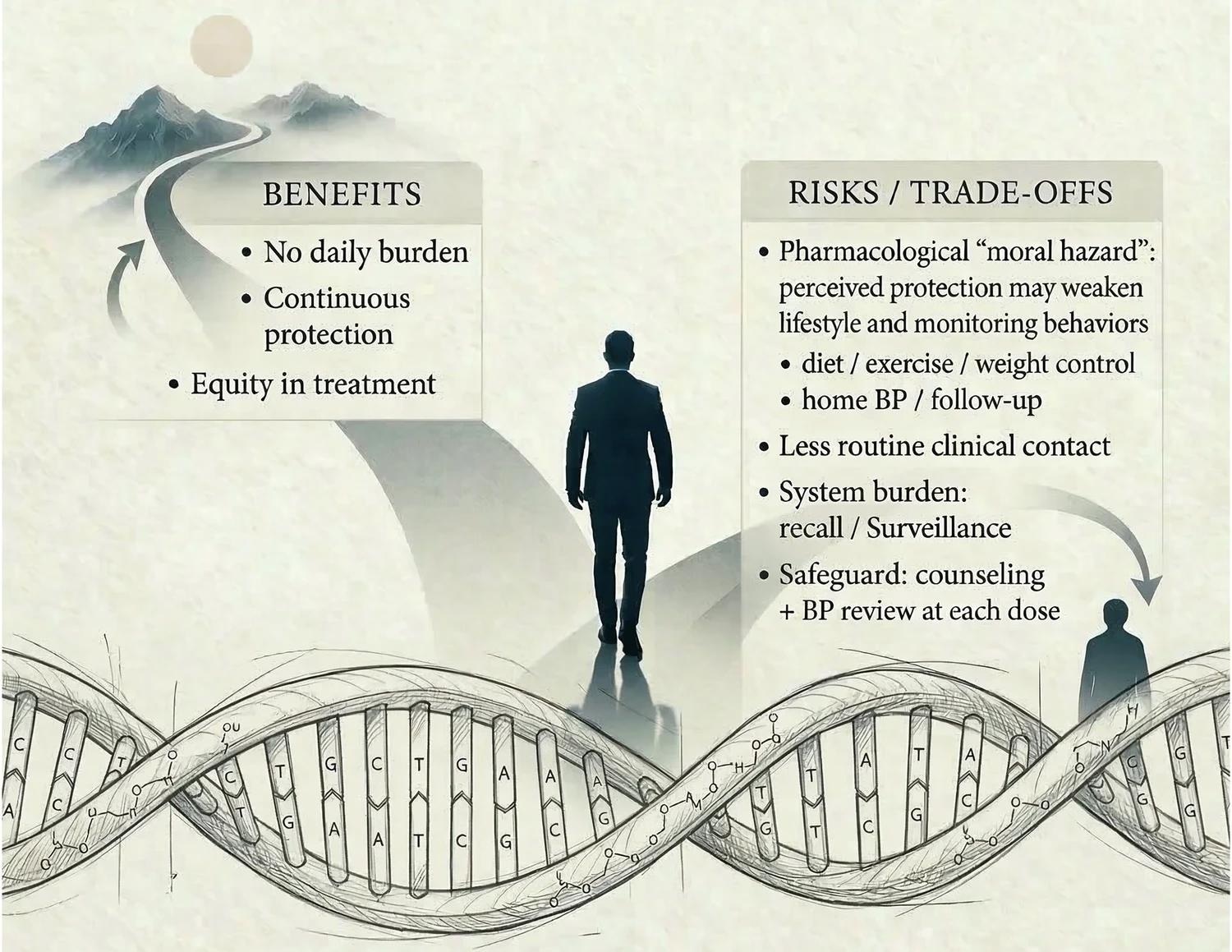
The Danger of “Pharmacological Moral Hazard”
With great convenience comes a new set of risks. Researchers have coined the term “pharmacological moral hazard” to describe a potential behavioral side effect of long-acting siRNA therapies.
The fear is that when a patient feels “totally secure” because of a twice-yearly injection, they may subconsciously de-prioritize the very lifestyle changes that preserve their heart healthy. This includes:
- Reducing sodium intake
- Managing body weight
- Engaging in regular physical activity
- Consistent home blood pressure monitoring
if patients only visit their doctor twice a year for an injection, hypertension may become less “visible.” Fewer clinical touchpoints could lead to a reduction in shared decision-making and a decline in routine monitoring.
Moving Beyond “Fire-and-Forget” Medicine
To prevent this, experts argue that health systems must resist a “fire-and-forget” mentality. A twice-yearly injection should not be the end of the conversation between a doctor and patient, but rather a “security floor.”
The goal is to turn each dosing visit into a high-value health checkpoint. Instead of a quick shot, these appointments should be used for:
- Lifestyle Reinforcement: Reviewing diet and exercise goals.
- Home BP Review: Analyzing data from home monitors to ensure stability.
- Medication Reconciliation: Ensuring all prescriptions are working in harmony.
- Safety Surveillance: Proactive monitoring for any adverse events.
The Future of Cardiovascular Protection
The promise of long-acting siRNA therapeutics lies in the democratization of health. By removing the “adherence trap,” People can potentially protect millions of people who struggle with the fragility of daily medication routines.
As we look toward the results of the ZENITH trial, the focus is shifting. The question is no longer just “Does the drug work?” but “Can this new model of care actually improve long-term cardiovascular outcomes?”
Frequently Asked Questions
What is siRNA therapy for hypertension?
Small-interfering RNA (siRNA) is a type of therapy that “silences” specific genes. In hypertension, drugs like zilebesiran target the production of angiotensinogen in the liver to lower blood pressure for months with a single dose.
Is zilebesiran a cure for high blood pressure?
No. It is a long-acting pharmacological intervention. While it stabilizes hemodynamics, it does not address the underlying lifestyle causes of hypertension.
What is “pharmacological moral hazard”?
It is the risk that patients may neglect healthy habits (like low-sodium diets or exercise) because they feel a false sense of total security from a long-acting medication.
How often would these injections be administered?
Based on current trials like KARDIA-1 and the planned ZENITH trial, dosing is being explored on a quarterly or biannual (twice-yearly) cadence.
Aim for to stay updated on the latest breakthroughs in cardiovascular health?
Join our community of health-conscious readers. Subscribe to our newsletter or leave a comment below to share your thoughts on the future of medicine!