Targeted Radiation Breakthrough Offers New Hope in Cancer Treatment
Researchers at The University of Texas MD Anderson Cancer Center have announced a significant step forward in precision cancer therapy with the development of a novel antibody designed to deliver targeted radiation directly to tumor cells. The findings, published in the journal Theranostics, detail a “first-in-class” antibody that zeroes in on a protein frequently overexpressed in a wide range of cancers, potentially opening doors to more effective and less damaging treatments.

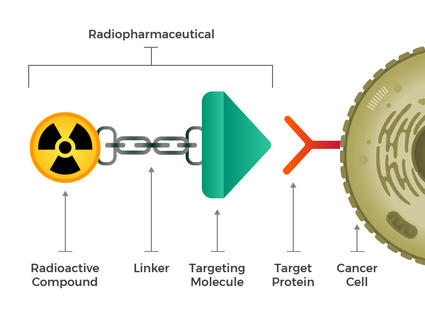
The new approach centers on a technique called theranostics – a field that, as MD Anderson explains, combines diagnostic imaging with targeted therapy. Initially, a low-dose radioactive substance, or radiotracer, is used to locate cancer cells using imaging scans like PET, and SPECT. This diagnostic phase informs a personalized treatment plan. Following diagnosis, a targeted radioactive medication is administered to bind to and destroy those identified cells [3].
This latest development focuses on the B7-H3 protein, a target that has proven challenging for researchers. The antibody developed by the MD Anderson team, led by David Piwnica-Worms, Seth Gammon, and Margie Sutton, is designed to specifically recognize and attach to the 4Ig-B7-H3 isoform of the protein, which is commonly found on tumor surfaces but less prevalent in healthy tissues [2]. Previous attempts to target B7-H3 have been hampered by the protein’s complex function and the existence of different isoforms, making it difficult to create therapies that accurately distinguish between cancerous and non-cancerous cells [2].
The ability to selectively target the 4Ig-B7-H3 isoform is a crucial advancement. The researchers explain that most prior therapies haven’t been able to differentiate it from the 2Ig-B7-H3 isoform, which circulates in the bloodstream [2]. This new antibody’s precision minimizes the risk of off-target effects, potentially reducing the side effects often associated with traditional radiation therapy.
MD Anderson is a leading center for theranostic treatments, offering a collaborative approach involving specialized teams of healthcare providers, including nurses, technologists, and pharmacists [1]. They currently offer Pluvicto®, a targeted radiopharmaceutical treatment for metastatic prostate cancer, demonstrating their commitment to advancing this field [3].
This research represents a significant step toward more personalized and effective cancer treatments, but further clinical trials will be necessary to fully evaluate its safety and efficacy in patients.
What challenges remain in translating this laboratory breakthrough into widely available cancer treatments?