Beyond the Microscope: The Recent Era of 3D Immune Mapping
For decades, scientists have known that cytotoxic T lymphocytes—the body’s specialized “killer” cells—are the frontline defense against cancer and infection. However, seeing these cells in action has always been a challenge. Traditional imaging often forces a trade-off: you can have high resolution, but you lose the three-dimensional context, or you preserve the structure but lose the detail.
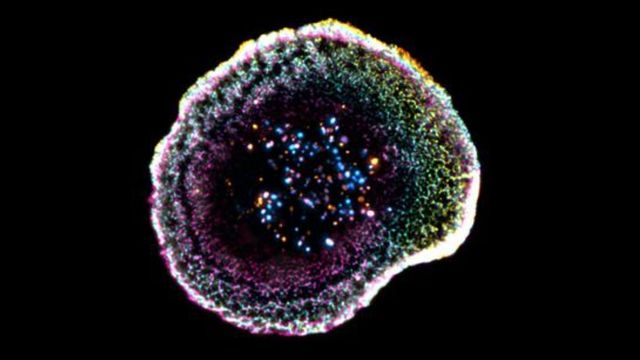
A breakthrough study from the University of Geneva (UNIGE) and the Lausanne University Hospital (CHUV), published in Cell Reports, has changed the game. By utilizing cryo-expansion microscopy (cryo-ExM), researchers can now visualize the molecular architecture of these cells in a near-native state, providing a blueprint for how the immune system actually destroys targets at the nanometer scale.
Decoding the “Immune Synapse”
The secret to a T cell’s efficiency is the “immune synapse.” This is a specialized exchange zone where the killer cell attaches to its target to release toxic molecules. The goal is surgical precision: destroy the cancerous cell without harming the healthy neighbors.
Using cryo-ExM, the research team discovered that at the point of contact, the membrane forms a “dome.” According to Florent Lemaître, a postdoctoral researcher at UNIGE and first author of the study, this structure “appears to be linked to adhesion interactions and to the internal organization of the cell.”
the study provided an unprecedented look at cytotoxic granules. These are the “weapons” of the T cell, and researchers found that their organization varies, with some containing one or more “cores” that concentrate the active molecules necessary for destruction.
From Lab Samples to Living Tumors
The true power of this imaging technique lies in its clinical application. Rather than relying solely on isolated cells in a petri dish, the team extended their approach to actual human tumor tissues.
This allowed them to directly observe T lymphocytes infiltrating tumors. Benita Wolf, Chief Resident and associate researcher at CHUV, notes that this capability allows scientists to “study immune responses directly in their clinical context and to better understand the mechanisms that determine their effectiveness.”
By moving from idealized lab environments to real-world tumor microenvironments, researchers can identify why some patients respond to immunotherapy whereas others do not.
How Precision Imaging Will Reshape Cancer Therapy
The ability to see the immune synapse in 3D isn’t just a win for microscopy; it is a roadmap for the future of immuno-oncology. By establishing a reference framework for how immune cells function, this research opens several doors for therapeutic innovation.
- Optimizing Drug Delivery: Understanding the “dome” structure of the immune synapse could lead to drugs that enhance the adhesion between T cells and cancer cells.
- Personalized Immunotherapy: By analyzing the cytotoxic machinery of a patient’s own T cells at the nanometer scale, doctors may eventually be able to predict the effectiveness of a treatment before it is administered.
- Overcoming Resistance: Visualizing why some T cells fail to form a functional synapse within a tumor can help scientists develop strategies to “unlock” the immune response in resistant cancers.
As we move toward more sophisticated molecular architecture studies, the focus is shifting from simply “activating” the immune system to “engineering” the precise physical interactions required for a kill.
The Future of Immuno-Oncology: What Comes Next?
The next frontier will likely involve combining cryo-expansion microscopy with real-time functional assays. While cryo-ExM provides a high-resolution “snapshot,” combining this with live-cell imaging could allow researchers to watch the “dome” form and the granules fire in real-time.

We are entering an era where the “invisible” mechanisms of the immune system are becoming visible. This shift from theoretical models to direct observation is essential for moving immunotherapy from a broad-spectrum approach to a precision science.
Frequently Asked Questions
What is cryo-expansion microscopy (cryo-ExM)?
It is an imaging technique that involves flash-freezing cells to preserve their structure and then physically expanding them using a hydrogel, allowing researchers to see internal cellular organization with extreme precision.
What is an immune synapse?
An immune synapse is the specialized contact zone between a T lymphocyte and a target cell (like a cancer cell) where the T cell releases molecules to destroy the target.
Why is 3D imaging better than traditional 2D imaging for T cells?
Traditional 2D imaging can alter fragile cellular structures and lose the spatial context. 3D imaging in a near-native state allows researchers to see the actual shape and organization of the cell, such as the “dome” formed during an attack.
What do you think about the future of precision medicine? Do you believe 3D imaging will be the key to curing resistant cancers? Let us know in the comments below or subscribe to our newsletter for the latest breakthroughs in medical science.