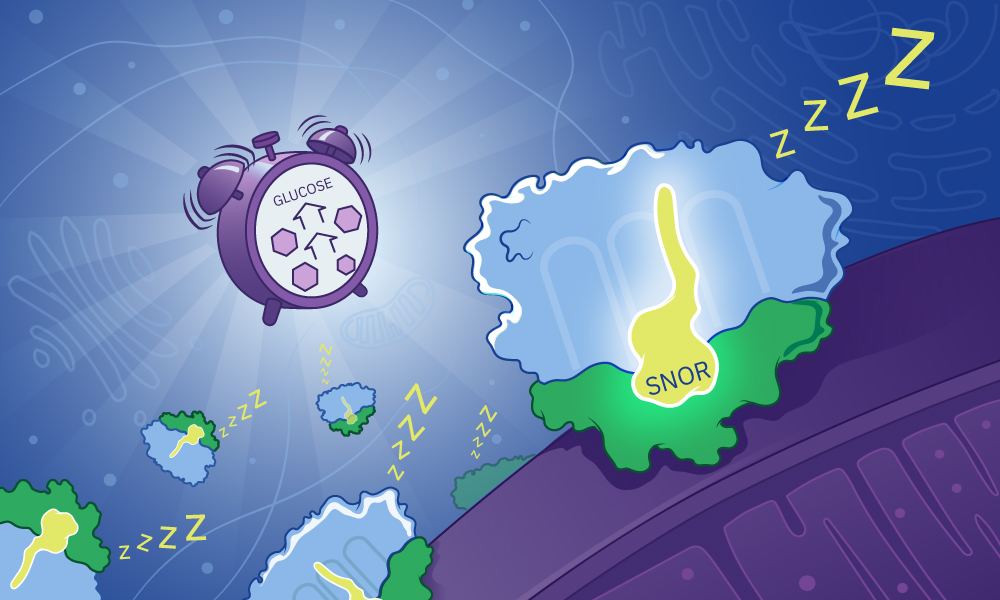
The “Sleeper” Cell Revolution: Beyond the SNOR Protein
For decades, biologists viewed cellular dormancy—or quiescence—as a passive state of “waiting.” When nutrients like glucose vanished, cells simply shut down to survive. However, the recent discovery of the SNOR protein by researchers at EMBL and the University of Virginia has flipped this narrative. Dormancy isn’t just a pause button; it’s a highly regulated biological choreography.
The discovery that SNOR acts as a molecular “all-clear” signal, triggering the rapid restart of protein synthesis within 30 minutes of nutrient availability, opens a door to a new era of metabolic engineering. We are moving from simply observing how cells survive stress to actively controlling how they wake up.
Cracking the Code of Cellular Hibernation
The ability of a cell to enter a dormant state and then precisely time its reactivation is a masterclass in evolutionary adaptation. In yeast and fungi, this mechanism ensures that the organism doesn’t waste precious energy trying to grow in a barren environment.

The future of this research lies in identifying the “upstream” triggers. While we know SNOR is the key that restarts the engine, we don’t yet know who turns the key. Understanding the signaling pathways that activate SNOR could allow scientists to artificially induce or prevent dormancy in various biological systems, leading to breakthroughs in metabolic health and cellular longevity.
From Yeast to Humans: The Cancer Connection
While SNOR was identified in yeast, the implications for human medicine are profound. One of the most challenging aspects of oncology is the existence of “cancer stem cells” that enter a dormant state, evading chemotherapy—which typically targets rapidly dividing cells.
If One can identify the human analogs of the SNOR protein, we could potentially develop therapies that:
- Lock cancer cells in dormancy: Preventing the “all-clear” signal from ever reaching the ribosome.
- Force “awakening”: Coaxing dormant cancer cells back into an active state to make them susceptible to traditional chemotherapy.
This shift toward targeting the transition between dormancy and activity, rather than the state itself, could redefine how we treat relapsing tumors.
The Tech Shift: Why “Visual Proteomics” Changes Everything
The discovery of SNOR wasn’t just a win for biology; it was a win for imaging technology. For years, structural biology relied on X-ray crystallography, which requires purifying proteins—essentially stripping them of their natural environment to see their shape.
The game-changer here is cellular cryo-electron tomography (cryo-ET). Instead of purifying the sample, researchers can now view 3D structures inside the cell. This “higher-definition” approach, combined with visual proteomics, allows scientists to spot “uncharacterised” proteins simply by looking at the map of the cell.
Seeing the Unseen in Real-Time
As cryo-ET resolution improves, we are entering an era of “molecular cinematography.” We will soon be able to watch proteins like SNOR attach to ribosomes in real-time as a cell senses a change in its environment. This will likely lead to the discovery of hundreds of other regulatory proteins that were previously invisible because they only appear during specific stress responses.
Future Horizons: Agriculture and Climate Survival
Beyond medicine, the ability to manipulate cellular “wake-up” calls has massive implications for global food security. As climate change creates more extreme weather patterns, crops face frequent periods of nutrient deprivation and drought.
By studying how spores germinate and how fungi manage dormancy via SNOR-like proteins, agricultural scientists could engineer “climate-resilient” crops. Imagine seeds that can remain dormant through an unprecedented drought but reactivate with lightning speed the moment the first rainfall occurs, maximizing the short growing windows available in volatile climates.
This intersection of structural biology and environmental science is where the next generation of sustainable agriculture will be born.
FAQ: Understanding Cellular Dormancy and SNOR
What exactly is the SNOR protein?
SNOR is a recently discovered protein in yeast that helps cells enter a dormant state during glucose starvation and, more critically, signals the ribosomes to restart protein synthesis once nutrients return.
How does cryo-ET differ from traditional microscopy?
Traditional methods often require purifying proteins, which removes their natural context. Cryo-ET allows scientists to see the 3D structure of proteins and organelles directly inside the cell, providing a more accurate “map” of cellular activity.
Can SNOR be used to treat human diseases?
While SNOR itself is found in fungi, the mechanism it uses is a blueprint. Finding similar “wake-up” proteins in humans could lead to new treatments for cancer and other diseases involving cellular quiescence.
Join the Conversation
Do you think the ability to control cellular dormancy will be the key to curing cancer, or is the risk of manipulating “sleeper cells” too high? We want to hear your thoughts on the future of visual proteomics!
Leave a comment below or subscribe to our newsletter for the latest breakthroughs in molecular biology.