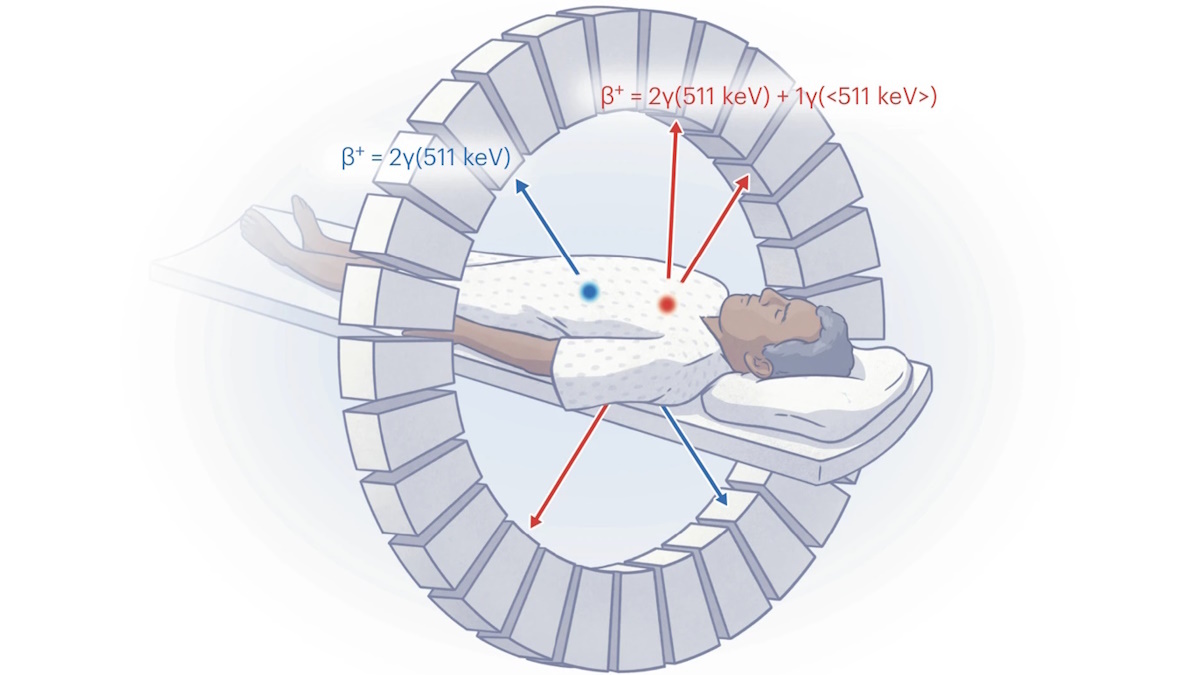
For decades, oncology has operated on a “map and blast” strategy. Doctors use imaging to find the tumor’s coordinates and then apply radiation to that specific area. While effective, this approach treats the tumor as a monolithic block of tissue, ignoring the chaotic, heterogeneous biological landscape inside the malignancy.
Enter multiplexed PET (Positron Emission Tomography). This isn’t just a marginal upgrade in image resolution. This proves a paradigm shift. By allowing clinicians to image multiple radiotracers simultaneously, we are moving from a black-and-white sketch of a tumor to a high-definition, multi-layered biological map.
Beyond the Single Tracer: The Power of Multiplexing
Standard PET scans typically rely on a single tracer, most commonly FDG (fluorodeoxyglucose), to highlight glucose metabolism. While useful, glucose uptake only tells one part of the story. A tumor might be metabolically active in one area but hypoxic (oxygen-starved) in another, or highly proliferative in a third.
Multiplexed PET allows us to see these different biological processes at the same time. Imagine being able to track oxygen levels, protein expression, and glucose metabolism in a single session. This provides a “molecular fingerprint” of the cancer, revealing exactly which parts of the tumor are the most aggressive and which are most resistant to treatment.
The Road to Biologically Individualized Radiotherapy
The ultimate goal of this technology is biologically individualized radiotherapy. Currently, radiation doses are often distributed uniformly across a tumor volume. However, we know that “hypoxic niches”—areas of the tumor with low oxygen—are notoriously resistant to radiation.
With multiplexed PET, oncologists can implement “dose painting.” By identifying the exact coordinates of these resistant niches using specific radiotracers, they can escalate the radiation dose to those stubborn areas while sparing the surrounding healthy tissue. This increases the probability of total tumor eradication while minimizing the devastating side effects of over-irradiation.
Real-World Implications: A Case Study in Precision
Consider a patient with a complex head-and-neck cancer. A standard scan shows a 4cm mass. A multiplexed scan, however, reveals that only 1cm of that mass is highly proliferative, while another 1cm is severely hypoxic. Instead of treating the entire 4cm mass with a medium dose, clinicians can target the proliferative and hypoxic zones with high-intensity beams, potentially reducing recurrence rates significantly.
The Convergence of Theranostics and AI
The future of this field lies in the marriage of multiplexed imaging and theranostics—a portmanteau of therapeutics and diagnostics. The idea is simple: use one isotope to find the target and a second, more powerful isotope to destroy it.
As we refine the ability to image multiple targets, we can develop “cocktails” of radiopharmaceuticals. For example, a patient could be imaged for both PSMA (Prostate-Specific Membrane Antigen) and GRPR (Gastrin-Releasing Peptide Receptor) expression. If the tumor expresses both, a dual-targeted therapy can be deployed, leaving the cancer with no escape route.
AI: The Great Decoder
The biggest hurdle in multiplexed PET is “signal crosstalk”—the difficulty of distinguishing between different tracers. This represents where Artificial Intelligence enters the fray. Machine learning algorithms are now being trained to recognize the unique decay signatures of different isotopes, effectively “unmixing” the image in real-time.
AI doesn’t just clean up the image; it predicts outcomes. By comparing a patient’s multiplexed biological map against thousands of previous cases, AI can suggest the optimal radiation dose and distribution, moving us closer to truly autonomous precision medicine.
Frequently Asked Questions
Q: Is multiplexed PET safe for the patient?
A: Yes. While it involves multiple tracers, the total radiation dose is carefully managed to remain within safety guidelines. In many cases, it is safer because it reduces the need for multiple separate scanning sessions.
Q: When will this be available in standard hospitals?
A: While currently primarily in the realm of advanced research and specialized academic centers, the transition to clinical practice is accelerating as AI decoding software becomes more standardized.
Q: How does this differ from a standard PET/CT scan?
A: A PET/CT combines metabolic data (PET) with anatomical data (CT). Multiplexed PET adds a third dimension: multi-biological data, allowing us to see different chemical processes simultaneously.
For more insights into the evolution of medical physics, explore our deep dives into next-generation radiopharmaceuticals and the future of proton therapy. You can similarly find peer-reviewed data on these advancements via the Nature Medical Imaging portal.
Join the Conversation
Do you believe molecularly-guided radiotherapy will become the gold standard in oncology within the next decade? Or is the complexity of implementation too high for widespread adoption?
Share your thoughts in the comments below or subscribe to our newsletter for the latest breakthroughs in precision medicine.