Unlocking the Kidney’s Secrets: Spatial Mapping and the Future of Diabetic Kidney Disease Treatment
A New Atlas of Kidney Complexity

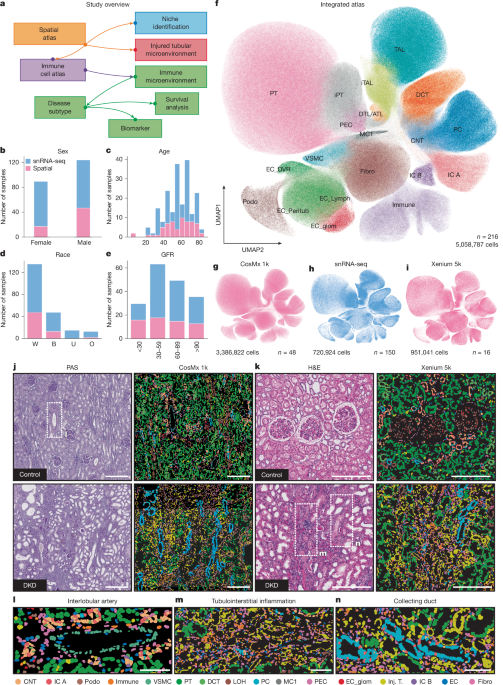
Diabetic kidney disease (DKD) remains a leading cause of kidney failure, but a recent study published in *Nature* has unveiled a detailed “atlas” of the kidney, offering unprecedented insight into the disease’s progression. Researchers at the University of Pennsylvania, utilizing advanced spatial transcriptomics technologies – Xenium and CosMx – alongside single-nucleus RNA sequencing, have created a computable map of kidney tissue architecture. This isn’t simply a visual representation. it’s a framework for understanding how different cell types and microenvironments interact in both healthy kidneys and those affected by DKD. The study, approved by the University of Pennsylvania institutional review board (IRB), utilized leftover kidney samples that had been irreversibly de-identified.
Identifying Key Microenvironments in DKD
The research pinpointed a profibrotic context that expands as DKD progresses, correlating with declining kidney function. Crucially, the team identified a B cell-predominant, tertiary lymphoid structure-like immune microenvironment. This suggests a previously underappreciated role for B cells in accelerating DKD progression. This discovery wasn’t just observational; researchers developed tissue biomarkers and a corresponding plasma protein panel capable of stratifying patients and improving risk prediction. These biomarkers hold promise for selecting patients for future B cell-targeted therapies.
The Power of Spatial Transcriptomics: Xenium and CosMx
The study’s success hinges on the application of cutting-edge spatial transcriptomics. Traditional RNA sequencing provides a snapshot of gene expression within a bulk tissue sample, losing crucial spatial information. Xenium and CosMx, however, allow researchers to measure gene expression *while* preserving the tissue’s architecture. Tissue sections were cut at 5 μm thickness and prepared according to manufacturer specifications, utilizing both universal cell characterization RNA probes and custom probes targeting specific genes. This detailed approach revealed nuanced differences in gene expression patterns within distinct kidney niches.
Beyond the Kidney: A Broader Impact on Precision Medicine
This research isn’t limited to DKD. The methodology – integrating multi-omics data with spatial information – is applicable to a wide range of diseases. The ability to create “computable tissue architecture” opens doors for non-invasive detection and personalized treatment strategies. The University of Pennsylvania’s Kidney Innovation Center, which houses a Human Kidney Tissue Biobank, is at the forefront of this effort, providing resources for researchers to advance kidney health and disease understanding.
Immune Cell Dynamics and the Role of B Cells
A significant finding centers on the immune response within the kidney. By integrating data from spatial transcriptomics with a pre-existing B cell atlas, researchers were able to refine the identification of B cell subtypes and their interactions with other cells. This revealed that specific B cell populations are concentrated in areas of inflammation and fibrosis, suggesting they actively contribute to disease progression. The study utilized a deep generative modelling framework using scVI and scANVI to integrate the data.
Predictive Biomarkers and Future Therapies
The identified plasma protein panel, derived from the spatial transcriptomic data, represents a potential breakthrough in DKD management. The study demonstrated that these biomarkers could improve risk prediction beyond traditional clinical models. This paves the way for more targeted interventions, potentially delaying or preventing the progression to kidney failure. Validation of these findings in the UK Biobank cohort further strengthens their clinical relevance.
Proteomic Insights: Unveiling IgA Interactions
Researchers also employed immune complex pull-down coupled with liquid chromatography-mass spectrometry (LC–MS) to identify proteins interacting with IgA in kidney tissues. This revealed a network of proteins involved in immune responses and inflammation, providing further clues about the mechanisms driving DKD.
The Rise of Spatial Proteomics and Multi-Omics Integration
The convergence of spatial transcriptomics, proteomics, and advanced computational analysis is transforming our understanding of complex diseases. Future research will likely focus on: * **Expanding Spatial Proteomics:** While spatial transcriptomics maps RNA expression, spatial proteomics directly measures protein levels, offering a more complete picture of cellular activity. * **Single-Cell Multi-Omics:** Combining multiple “omics” layers (genomics, transcriptomics, proteomics, metabolomics) at the single-cell level will provide unprecedented resolution. * **Artificial Intelligence and Machine Learning:** AI algorithms will be crucial for analyzing the vast datasets generated by these technologies and identifying novel biomarkers and therapeutic targets. * **Personalized Medicine Approaches:** Tailoring treatments based on an individual’s unique molecular profile and spatial organization of their kidney tissue.
FAQ
**Q: What is spatial transcriptomics?** A: It’s a technology that allows researchers to measure gene expression while preserving the spatial context of cells within a tissue. **Q: How does this research support DKD patients?** A: It identifies new biomarkers for risk prediction and potential targets for B cell-targeted therapies. **Q: What is a “computable tissue architecture”?** A: It’s a digital map of the kidney that allows researchers to analyze how different cell types and microenvironments interact. **Q: Is this research applicable to other diseases?** A: Yes, the methodology can be applied to a wide range of diseases beyond DKD. **Did you know?** The human kidney contains millions of nephrons, each a complex filtering unit. Understanding the spatial organization of these nephrons is crucial for understanding kidney function and disease. **Pro Tip:** Stay informed about the latest advancements in spatial biology by following leading research institutions and journals in the field. This research represents a significant step forward in our understanding of DKD and highlights the transformative potential of spatial biology for precision medicine. As technology continues to advance, we can expect even more detailed and insightful maps of the human body, leading to more effective treatments and improved patient outcomes.