The Evolution of White Matter Research: From Lesion Models to Spatial Omics
The landscape of neuroscience is shifting toward an era of unprecedented precision. Recent methodologies in studying white matter demyelination—specifically within the central posterior cortex (CCP), corpus callosum, and cingulum—reveal a trend toward integrating real-time functional monitoring with high-resolution genetic mapping.
By utilizing sophisticated models such as EtBr (Ethidium Bromide) and lysolecithin, researchers can now induce focal demyelination to study the brain’s capacity for repair. The focus is no longer just on whether a lesion occurs, but on the cellular choreography that follows.
Precision Mapping via Spatial Transcriptomics (DBiT-seq)
One of the most significant trends in neural research is the move from bulk RNA sequencing to spatial transcriptomics. Even as bulk RNA-seq provides a general overview of gene expression, DBiT-seq (Deterministic Barcoding in Tissue) allows for the mapping of transcriptomes directly onto the tissue’s physical structure.
This approach enables the delineation of specific pixel clusters, such as those expressing Calb1 (calbindin) in the inferior olive (IO), or identifying microglia within a lesion using marker genes like Aif1, Maf, Spi1, and Csf1r. This level of detail transforms our understanding of how different cell types interact at the exact site of injury.
For more information on how institutional frameworks support this high-level research, you can explore the University of Cambridge’s resources on staff and visitor immigration, which facilitate the global exchange of scientific expertise.
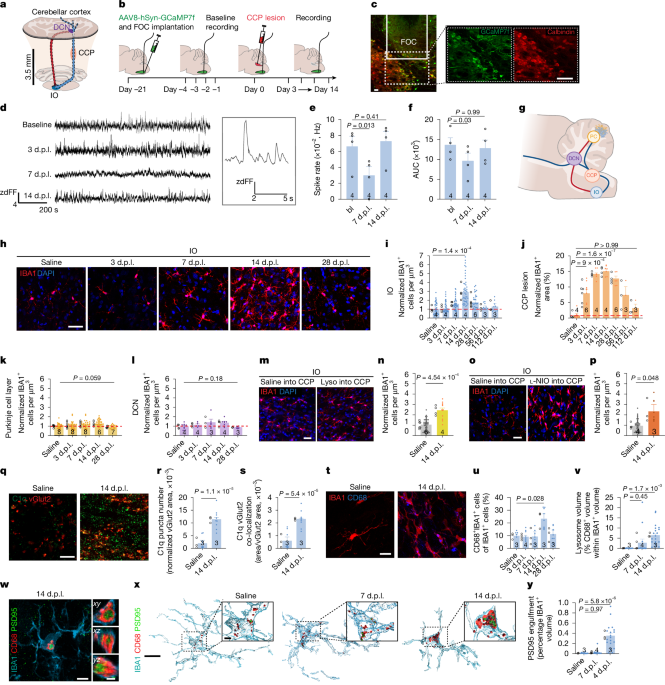
The Microglial Pivot: From Inflammation to Synaptic Engulfment
Current research is deeply investigating the role of microglia—the brain’s resident immune cells—not just as inflammatory agents, but as active participants in synaptic remodeling. Advanced imaging techniques now allow for the quantification of synaptic engulfment.
By using markers like IBA1 for microglia and PSD95 or bassoon for synaptic proteins, scientists can calculate an “engulfment index.” This measures the volume of synaptic proteins contained within CD68+ lysosomes inside microglia. This trend highlights a move toward understanding how the removal of synaptic material influences the environment for remyelination.
the use of Csf1r blockers like PLX5622 for local microglial depletion allows researchers to isolate the specific impact of these cells on the remyelination process, providing a clearer picture of their necessity in nerve repair.
Real-Time Functional Monitoring with Fibre Photometry
The ability to watch the brain “work” in real-time is expanding through fibre photometry. The trend is moving toward using more sensitive calcium indicators, such as GCaMP8m, which offers a greater signal-to-noise ratio compared to earlier versions like GCaMP7f.
Beyond calcium, the deployment of GRABATP1.0—a modified human P2Y receptor—allows for the real-time sensing of extracellular ATP. This enables researchers to link chemical releases to specific neuronal activity patterns during the exploration of a home-cage environment, bridging the gap between molecular changes and behavioral states.
Mitochondrial Morphology as a Biomarker for Health
There is a growing emphasis on the “energetics” of the neuron. By genetically labeling mitochondria (e.g., using MitoDsRed), researchers are now analyzing volume-weighted mitochondrial sphericity.
A sphericity value closer to 1 indicates a more spherical morphology, which can serve as a critical indicator of mitochondrial health and function within calbindin+ neuronal cell bodies. Analyzing these dynamics at neuron-microglia junctions provides a new window into how metabolic support is managed during white matter injury.
For those interested in the regulatory side of such animal studies, the GOV.UK Student Sponsor Guidance outlines the premises and duties associated with high-level research institutions.
AI-Driven Histology and Remyelination Analysis
The manual ranking of remyelination is being replaced by AI-based segmentation. Using DenseNet networks, researchers can now automatically quantify remyelinated versus non-remyelinated regions within focal white matter lesions.
This AI approach, validated by electron microscopy and g-ratio calculations, removes human bias and allows for the analysis of hundreds of annotations across multiple lesions with high classification accuracy (cross-entropy < 0.001). This represents a broader trend toward the “digitization” of pathology.
Frequently Asked Questions
What is the purpose of using EtBr in white matter models?
EtBr (Ethidium Bromide) is used to induce focal white matter demyelination, allowing researchers to study how the brain responds to the loss of myelin and the subsequent attempts at regeneration.

How does DBiT-seq differ from standard RNA sequencing?
Unlike standard bulk RNA-seq, which averages gene expression across a whole tissue sample, DBiT-seq provides spatial resolution, mapping exactly where specific genes are being expressed within the tissue architecture.
What is the role of OPCs in these studies?
Oligodendrocyte Progenitor Cells (OPCs) are highly proliferating progenitors essential for remyelination. Research often focuses on why these cells sometimes fail to regenerate myelin, using markers like EdU to track their proliferation.
How is the “engulfment index” calculated?
It is calculated by taking the volume of synaptic proteins (like PSD95) found within CD68+ lysosomes and dividing it by the total IBA1 volume of the microglial cell, expressed as a percentage.
What do you think is the most promising technology for treating white matter injury? Share your thoughts in the comments below or subscribe to our newsletter for more deep dives into neuroscience!