The Future of Cancer Immunotherapy: In Vivo CAR-T Cell Therapy Gains Momentum
The landscape of cancer treatment is undergoing a dramatic shift, with immunotherapy rapidly emerging as a cornerstone of care. Recent breakthroughs, particularly in CAR-T cell therapy, are offering recent hope to patients battling previously intractable cancers. Now, a potentially game-changing approach – in vivo CAR-T cell generation – is showing promising early results, hinting at a future where cancer treatments are faster, more accessible, and potentially less toxic.
What is In Vivo CAR-T Cell Therapy?
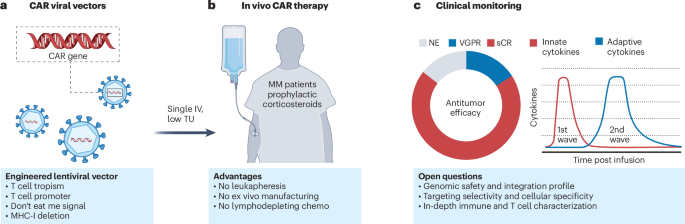
Traditional CAR-T cell therapy involves extracting a patient’s T cells, genetically engineering them to express a Chimeric Antigen Receptor (CAR) that targets cancer cells, and then infusing these modified cells back into the patient. This process is complex, expensive, and requires specialized facilities. In vivo CAR-T therapy, however, aims to bypass these steps by delivering a gene-encoding CAR directly into the patient’s body. This prompts the patient’s own T cells to become CAR-T cells within the body, eliminating the necessitate for external manipulation.
Early Data Shows Promise in Multiple Myeloma
A recent study published in Nature Medicine provides the first clinical data on this innovative approach, focusing on patients with relapsed or refractory multiple myeloma (MM). The research utilized an experimental lentivirus, ESO-T01, to deliver anti-BCMA CAR genes directly to patients. While the trial is still in its early stages, the data reveals the feasibility of generating CAR-T cells directly within the patient. This represents a significant step towards streamlining the CAR-T process.
Addressing the Challenges of Traditional CAR-T Therapy
Current CAR-T therapies, while effective, are not without limitations. High costs, logistical complexities, and potential toxicities – such as cytokine release syndrome (CRS) and neurotoxicity – can restrict access and create challenges for patients. In vivo CAR-T therapy offers a potential solution to many of these issues. By simplifying the manufacturing process, it could dramatically reduce costs and make this life-saving treatment available to a wider patient population.
Beyond Multiple Myeloma: Expanding the Potential
While the initial research focuses on multiple myeloma, the potential applications of in vivo CAR-T therapy extend far beyond this blood cancer. Researchers are exploring its use in solid tumors and other hematological malignancies. The ability to generate CAR-T cells directly within the tumor microenvironment could prove particularly advantageous in overcoming the challenges posed by solid tumors, where CAR-T cell penetration and persistence are often limited.
Did you know? BCMA (B cell maturation antigen) is a protein found on the surface of myeloma cells, making it an ideal target for CAR-T therapy.
Safety Concerns and Future Research
The Nature Medicine study also highlighted significant safety concerns. All patients in the early trial experienced serious toxicities. This underscores the need for careful dose optimization and further research to mitigate these risks. Future studies will focus on refining the delivery methods, improving the specificity of the CAR, and developing strategies to manage potential side effects.
Pro Tip: Understanding the tumor microenvironment is crucial for optimizing CAR-T cell therapy. Factors like immune suppression and antigen loss can impact treatment efficacy.
The Evolving Landscape of CAR-T Cell Therapies
The field of CAR-T cell therapy is rapidly evolving. Researchers are exploring new CAR designs, targeting different antigens, and combining CAR-T therapy with other treatments, such as immunomodulatory drugs. The development of in vivo CAR-T therapy represents another exciting advancement, potentially paving the way for a new generation of immunotherapies.
FAQ
Q: What is the main difference between traditional and in vivo CAR-T therapy?
A: Traditional CAR-T therapy requires T cells to be modified outside the body, while in vivo CAR-T therapy generates CAR-T cells directly within the patient’s body.
Q: Is in vivo CAR-T therapy safer than traditional CAR-T therapy?
A: Early data suggests potential safety concerns, and further research is needed to assess and mitigate these risks.
Q: What types of cancer could benefit from in vivo CAR-T therapy?
A: Initial research focuses on multiple myeloma, but the therapy has potential applications in other hematological malignancies and solid tumors.
Q: How much does CAR-T therapy cost?
A: Traditional CAR-T therapies are very expensive, often exceeding $300,000 per treatment. In vivo CAR-T therapy aims to reduce these costs by simplifying the manufacturing process.
Want to learn more about the latest advancements in cancer treatment? Explore our other articles on immunotherapy and stay informed about the future of cancer care.