The Future of Reproductive Biology: Insights from Pronuclear Dynamics and Genome Editing
Recent research, meticulously detailed in a study published in Nature, sheds light on the intricate processes governing early mammalian development. The work, conducted with a focus on mouse models, delves into the mechanics of pronuclear formation, genome organization and the impact of cytoplasmic volume on successful fertilization. These findings aren’t just academic exercises; they point towards potential advancements in assisted reproductive technologies (ART) and a deeper understanding of early embryonic loss.
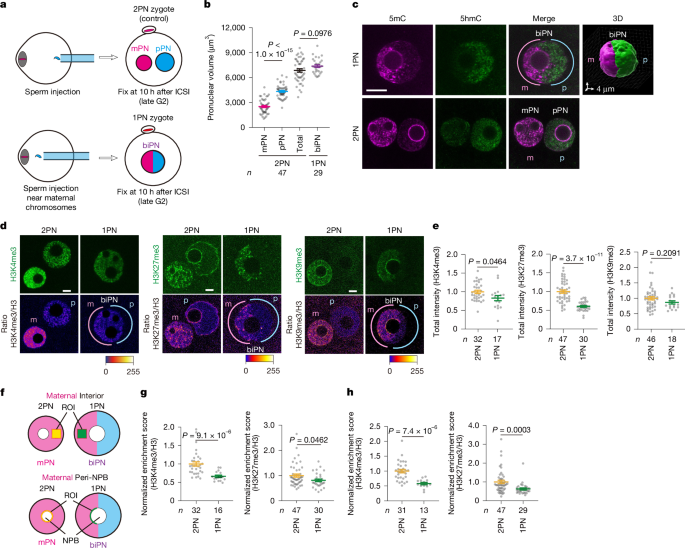
Unraveling the Mysteries of Pronuclear Formation
The study meticulously examined the formation and behavior of pronuclei – the structures containing the maternal and paternal genomes after fertilization. Researchers utilized advanced imaging techniques, including live-cell imaging and three-dimensional analysis, to observe these processes in real-time. They discovered that pronuclear volume is a critical factor influencing developmental success. Manipulating cytoplasmic volume, through techniques like cytoplasmic removal and oocyte doubling, directly impacted pronuclear formation and subsequent embryo development.

This research builds upon previous work identifying the C57BL/6 mouse strain as a cornerstone of biomedical research due to its genetic homogeneity. The study utilized several strains – C57BL/6, JF1/Ms, and BDF1 – alongside transgenic lines, highlighting the importance of precise genetic control in these experiments.
The Role of Histone Modifications and Genome Organization
A key aspect of the research focused on histone modifications – chemical alterations to DNA packaging proteins that influence gene expression. Specifically, the team investigated the roles of H3K4me3, H3K27me3, and H3K27ac in establishing proper genome organization within the pronuclei. They found that these modifications are dynamically regulated and play a crucial role in preparing the genome for subsequent embryonic development.
Using a technique called CATCH-seq, an ultra-low-input chromatin immunoprecipitation method, researchers were able to analyze histone modifications with unprecedented sensitivity. This allowed them to identify allele-specific differences in histone modification patterns, suggesting a complex interplay between maternal and paternal genomes during early development.
Implications for Assisted Reproductive Technologies
The findings have significant implications for ART, such as in vitro fertilization (IVF) and intracytoplasmic sperm injection (ICSI). The study demonstrates that optimizing cytoplasmic volume and ensuring proper pronuclear formation can improve embryo quality and increase the chances of successful implantation.

“Our work suggests that simply injecting sperm into an oocyte isn’t enough,” explains the research. “The cellular environment, particularly the amount of cytoplasm, plays a critical role in supporting proper genome organization and development.” This could lead to refinements in ICSI protocols, potentially improving outcomes for couples undergoing fertility treatment.
Beyond IVF: Understanding Early Embryonic Loss
Early embryonic loss is a major challenge in both natural conception and ART. The research suggests that disruptions in pronuclear formation, genome organization, or histone modification patterns could contribute to these losses. By identifying the key factors involved in successful early development, researchers hope to develop strategies to prevent or mitigate these issues.

The study also explored the impact of manipulating the number of pronuclei, creating doubled or halved zygotes. These experiments provided valuable insights into the essential components required for normal development, further illuminating the causes of early embryonic failure.
The Future of Genome Editing and Early Development
The study’s findings dovetail with advancements in genome editing technologies like CRISPR-Cas9. A deeper understanding of pronuclear dynamics and genome organization will be crucial for safely and effectively applying these technologies to correct genetic defects in embryos.
Researchers are also exploring the potential of using RNA sequencing to assess the quality of embryos before implantation. Single-embryo RNA-seq, employed in this study, allows for a comprehensive analysis of gene expression patterns, providing a more accurate assessment of developmental potential.
FAQ
Q: What are pronuclei? A: Pronuclei are the structures containing the maternal and paternal genomes after fertilization, before they fuse to form the zygote nucleus.

Q: Why is cytoplasmic volume important? A: Cytoplasmic volume influences pronuclear formation, genome organization, and overall embryo development.
Q: How can this research improve IVF? A: By optimizing cytoplasmic volume and ensuring proper pronuclear formation, IVF success rates may be improved.
Q: What is CATCH-seq? A: CATCH-seq is a highly sensitive technique for analyzing chromatin structure and histone modifications in very small samples, like single cells or pronuclei.
Q: What role do histone modifications play? A: Histone modifications regulate gene expression and are crucial for establishing proper genome organization during early development.
Pro Tip
When considering ART, discuss with your physician the latest advancements in embryo selection and quality assessment techniques. A comprehensive evaluation of embryo health can significantly increase your chances of a successful pregnancy.
Did you recognize?
The C57BL/6 mouse strain, used extensively in this research, has over 20 substrains worldwide, each with potentially different phenotypic characteristics. This highlights the importance of carefully selecting the appropriate strain for research purposes.
This research represents a significant step forward in our understanding of early mammalian development. As technology continues to advance, we can expect even more detailed insights into the intricate processes that govern life’s beginnings, paving the way for improved reproductive technologies and a reduction in early embryonic loss.