Predicting the Unpredictable: New Tool Offers Hope in Smoldering Multiple Myeloma Monitoring
Dana-Farber Cancer Institute researchers have unveiled PANGEA-SMM, a new online tool designed to more accurately predict when smoldering multiple myeloma (SMM) will progress to active cancer. This development marks a significant step forward in personalized cancer care, offering the potential to refine treatment strategies and reduce unnecessary interventions.
Understanding Smoldering Multiple Myeloma and the Need for Better Prediction
Smoldering multiple myeloma is a precursor condition to active multiple myeloma. Not everyone with SMM will develop the full-blown cancer, making it challenging to determine who requires immediate treatment and who can safely be monitored. Current predictive tools often rely on a single snapshot of a patient’s lab results, potentially missing crucial information about the disease’s trajectory.
How PANGEA-SMM Works: A Dynamic Approach
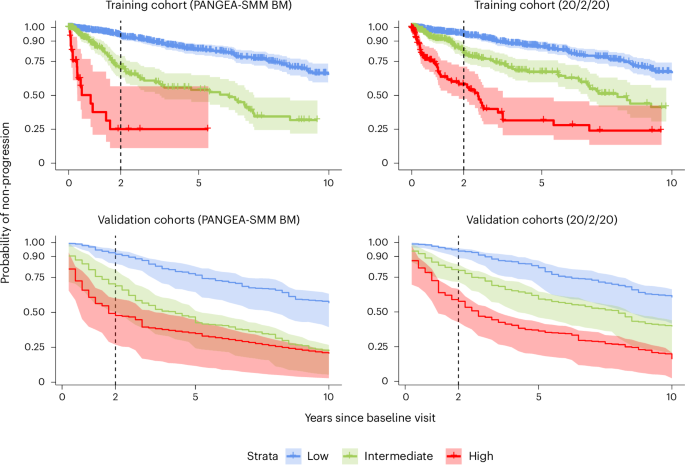
PANGEA-SMM distinguishes itself by incorporating dynamic biomarkers – tracking changes in lab results over time. The tool analyzes key indicators like M-protein concentration, sFLC ratio, creatinine and hemoglobin. By observing the speed and direction of these changes, PANGEA-SMM aims to identify high-risk patients who would benefit from early treatment while sparing those with stable disease from unnecessary interventions. The tool is accessible globally, leveraging routinely used lab measures.
A Collaborative Effort: Data from Multiple Institutions
The development of PANGEA-SMM involved a substantial cohort of patients. The initial training cohort comprised 1,031 individuals diagnosed with SMM at Dana-Farber Cancer Institute. To ensure robustness, the model was validated using data from five independent cohorts across six international centers, including institutions in Greece, the UK, Germany, Italy, and Spain. This multi-institutional approach strengthens the reliability and generalizability of the tool.
Rigorous Validation and Performance
The study, published in Nature Medicine, underwent a rigorous validation process. Researchers assessed ranking accuracy, risk stratification, and calibration of the PANGEA-SMM models. Even with less frequent observations – for example, one visit per year – the tool maintained comparable performance. The research team also developed an open-access web application allowing users to evaluate the tool’s performance on training data and a subset of validation cohorts.
Ethical Considerations and Data Handling
The study was approved by the Dana-Farber/Harvard Cancer Center institutional review board. Due to the retrospective nature of the data and minimal risk to patients, a waiver of informed consent was granted for the initial cohort. However, informed consent was obtained from patients in several validation cohorts, adhering to ethical guidelines and local regulations.
The Future of SMM Monitoring: Personalized Risk Assessment
PANGEA-SMM represents a shift towards more personalized risk assessment in SMM management. The tool’s ability to dynamically track biomarker changes offers a more nuanced understanding of disease progression than traditional static models. This could lead to more informed treatment decisions, potentially delaying or avoiding treatment for patients at low risk and initiating therapy earlier for those at high risk.
Open-Access Tools Empowering Clinicians and Patients
Dana-Farber has made PANGEA-SMM freely available through an open-access web application. This accessibility is crucial for widespread adoption and impact, allowing clinicians worldwide to utilize the tool in their practice. A clinical calculator is also available, enabling users to input individual patient data and receive personalized risk assessments.
Pro Tip:
Regular monitoring of biomarker trends, as facilitated by tools like PANGEA-SMM, is key to effective SMM management. Discuss the potential benefits of dynamic risk assessment with your healthcare provider.
Frequently Asked Questions
Q: What is smoldering multiple myeloma?
A: It’s a precursor condition to active multiple myeloma, where abnormal plasma cells are present but don’t yet cause significant symptoms.
Q: How does PANGEA-SMM differ from existing tools?
A: PANGEA-SMM analyzes changes in biomarkers over time, providing a more dynamic and accurate prediction of disease progression.
Q: Is PANGEA-SMM available to all patients?
A: Yes, the tool is open-access and available online to clinicians worldwide.
Q: What data is needed to use PANGEA-SMM?
A: The tool requires data on M-protein concentration, sFLC ratio, creatinine, hemoglobin, and optionally, BMPC.
Did you know?
The PANGEA project utilized the largest cohort to date for characterizing the transition from SMM to MM, comprising over 1,000 patients in the training cohort alone.
To learn more about PANGEA-SMM and access the tool, please visit the Dana-Farber Cancer Institute website. Consider discussing this new technology with your physician to explore its potential benefits for your individual situation.