Precision Medicine Gains Momentum in Pancreatic and Biliary Tract Cancers: A New Era of Targeted Therapies
The landscape of treatment for pancreatic and biliary tract cancers is undergoing a significant shift, moving away from broad-spectrum approaches towards highly personalized strategies. Recent clinical trials, like the POLAR and related studies, are demonstrating the potential of combining immunotherapy with targeted therapies, particularly in patients with specific genetic mutations. This article delves into the latest findings and explores the future direction of these advancements.
Understanding the Role of HRD and Biomarkers
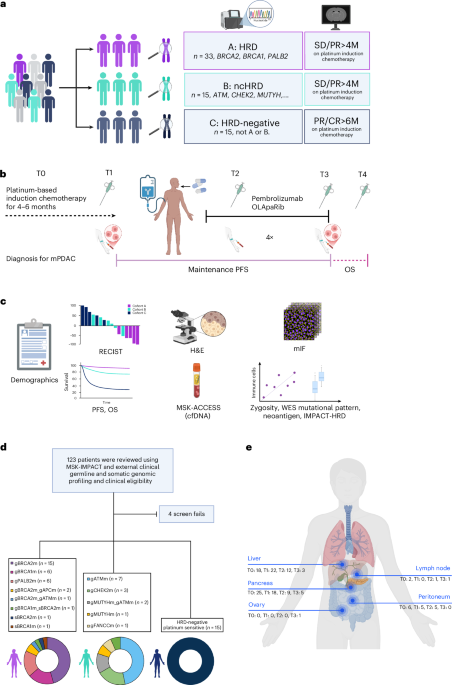
A key focus of current research is identifying patients who will respond best to specific treatments. The POLAR trial, evaluating pembrolizumab plus olaparib in metastatic pancreatic cancer, stratified participants into three cohorts based on Homologous Recombination Deficiency (HRD) status. Cohort A, encompassing patients with mutations in BRCA1/2 or PALB2, showed promising, though not statistically significant, objective response rates. Further analysis revealed that patients with specific mutations, like BRCA2 and PALB2, tended to experience more prolonged progression-free survival than those with BRCA1 mutations.
Beyond BRCA mutations, the study as well examined the impact of mutations in non-core HRD genes like ATM and CHEK2. While the overall response rates were lower in these groups, the research highlights the importance of comprehensive genomic profiling to identify potential candidates for targeted therapies. The POLAR trial also investigated the role of circulating tumor DNA (ctDNA) dynamics, finding that minimal residual disease, indicated by low variant allele frequency, correlated with durable clinical benefit.
Biliary Tract Cancer: Pembrolizumab and Olaparib Combination Shows Promise
Similar strategies are being explored in biliary tract cancer. A phase II study combining pembrolizumab and olaparib demonstrated an objective response rate of 15.4% and a disease control rate of 53.8% in patients with advanced disease. Median progression-free survival was 5.45 months, and overall survival reached 7.21 months. Notably, patients with IDH1 mutations or HRR deficiencies appeared to benefit the most from this combination, suggesting a potential rechallenge with immunotherapy for these subgroups.
The Importance of Tumor Microenvironment and Immune Infiltration
Recent research emphasizes the critical role of the tumor microenvironment in treatment response. Studies have shown that tumors with higher levels of tumor-infiltrating lymphocytes (TILs) and increased frameshift indel mutations tend to respond better to immunotherapy. The POLAR trial’s analysis of tumor samples revealed that HRD tumors exhibited a more immunogenic mutational landscape, with higher levels of neoantigens and greater immune cell infiltration compared to non-HRD tumors.
Safety and Tolerability
The combination of pembrolizumab and olaparib generally demonstrated a manageable safety profile. The POLAR trial reported no grade 4 or 5 treatment-related adverse events, with the most common grade 3 events being anemia and abdominal infection. Immune-related adverse events, such as colitis and pneumonitis, were observed but were generally manageable.
Future Directions and Emerging Trends
The data from these trials points towards several key areas for future research:
- Expanded Biomarker Testing: Wider adoption of comprehensive genomic profiling to identify patients with HRD mutations and other predictive biomarkers.
- Novel Combinations: Investigating new combinations of immunotherapy with targeted therapies, potentially including PARP inhibitors, to overcome resistance mechanisms.
- ctDNA Monitoring: Utilizing ctDNA analysis to monitor treatment response and detect early signs of disease progression.
- Personalized Immunotherapy: Developing personalized immunotherapy approaches based on the individual patient’s tumor mutational burden and immune microenvironment.
FAQ
Q: What is HRD?
A: Homologous Recombination Deficiency is a genetic defect that impairs the cell’s ability to repair DNA, making it more susceptible to certain targeted therapies.
Q: What are PARP inhibitors?
A: PARP inhibitors are drugs that block an enzyme involved in DNA repair, and are particularly effective in tumors with HRD mutations.
Q: What is ctDNA?
A: Circulating tumor DNA is DNA released by cancer cells into the bloodstream, which can be analyzed to monitor treatment response and detect mutations.
Q: Are these treatments available to all patients?
A: Currently, these treatments are typically reserved for patients with specific genetic mutations and advanced disease. Access may vary depending on location and insurance coverage.
Did you understand? Patients with BRCA2 mutations in the POLAR trial demonstrated numerically similar PFS and OS, but longer than those with BRCA1 mutations.
Pro Tip: Discuss comprehensive genomic profiling with your oncologist to determine if you are a candidate for targeted therapies.
Stay informed about the latest advancements in pancreatic and biliary tract cancer treatment. Explore additional resources from leading cancer organizations and research institutions to learn more about personalized medicine and clinical trials.