The Future of Cancer Treatment: Beyond Approved Labels
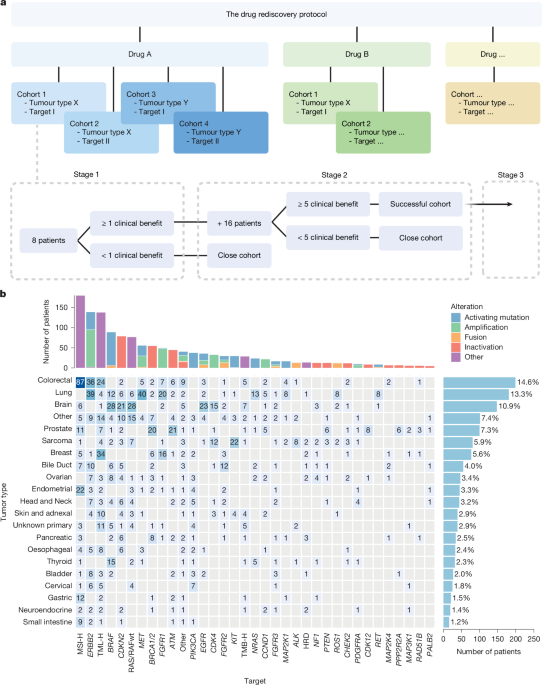
For years, cancer treatment has largely followed a rigid path: diagnosis, standard therapy, and then… limited options. But a paradigm shift is underway, fueled by trials like the Dutch DRUP (Drug Rediscovery Protocol) and a growing understanding of the power of genomic profiling. DRUP, which has now included over 1,600 patients, demonstrates that existing drugs, used “off-label” – meaning for cancers they weren’t originally approved to treat – can offer substantial benefit when matched to a patient’s unique genetic makeup.
Unlocking Potential in Existing Drugs
The core principle behind DRUP is simple: cancer isn’t defined by where it starts, but by what drives its growth at a molecular level. By analyzing a tumor’s DNA for specific alterations – mutations, deletions, and amplifications – doctors can identify drugs approved for other cancers that might be effective. This approach addresses a critical gap in precision oncology: the scarcity of treatments tailored to the diverse mutational landscapes found in less common cancers.
Recent findings, published in Nature, show that roughly one-third of patients in DRUP experienced clinical benefit, with nearly 16% achieving an objective response and 7% becoming exceptional responders – remaining progression-free for at least two years. These results are comparable to those seen in other precision oncology trials like NCI-MATCH and MyPathway, validating the potential of this strategy.
The Risks of Off-Label Utilize Without Oversight
Despite the promise, off-label drug use isn’t without its dangers. Currently, many oncologists prescribe these treatments outside of clinical trials, lacking the systematic evaluation of efficacy and toxicity. This can expose patients, often already weakened by previous treatments, to serious side effects – with nearly 28.4% of DRUP patients experiencing grade 3 or higher treatment-related adverse events – and create unequal access to potentially life-saving therapies.
the financial burden can be significant. High drug prices, inconsistent reimbursement policies, and the cost of managing toxicities all contribute to the overall expense. Structured initiatives like DRUP, along with similar trials in Canada (CAPTUR) and across Europe (PCM4EU and PRIME-ROSE), are crucial for mitigating these risks by providing controlled access and generating vital data.
Key Factors Influencing Treatment Success
DRUP’s data reveals several factors that influence the success of off-label therapies. Drugs with a strong biological rationale and prior clinical evidence of activity – those ranked highly by systems like the ESMO Scale for Clinical Actionability of molecular Targets (ESCAT) and OncoKB – tend to perform better. For example, treatments targeting BRAF p.V600E mutations, MSI-H, and TMB-H have shown promising results.

Interestingly, administering these therapies earlier in the disease course, before patients have undergone multiple lines of treatment, appears to improve outcomes. However, the relevance of tissue context – the original location of the cancer – remains a complex issue. While some molecular alterations respond consistently across cancer types, others exhibit tissue-specific effects, highlighting the need for nuanced approaches.
The Role of International Collaboration
One significant hurdle to progress is the rarity of certain genetic alterations and cancer subtypes. To overcome this, international collaboration is essential. The success of DRUP has inspired similar trials across Europe, and projects like PCM4EU and PRIME-ROSE are fostering data sharing and harmonized protocols to accelerate enrollment and validate findings.
Looking Ahead: What’s Next for Precision Oncology?
The future of cancer treatment lies in a more refined and data-driven approach. Several key trends are emerging:
- Enhanced Molecular Diagnostics: Wider access to comprehensive genomic profiling will be crucial for identifying patients who might benefit from off-label therapies.
- Artificial Intelligence (AI) and Machine Learning: AI algorithms can analyze vast datasets to predict drug sensitivity and identify novel therapeutic targets.
- Liquid Biopsies: These non-invasive blood tests can detect circulating tumor DNA, providing a real-time snapshot of a tumor’s genetic makeup and response to treatment.
- Real-World Evidence (RWE): Collecting and analyzing data from routine clinical practice will complement data from clinical trials, providing a more comprehensive understanding of treatment effectiveness.
FAQ
Q: What is “off-label” drug use?
A: Using a drug for a condition it wasn’t originally approved for by regulatory agencies.
Q: Is off-label drug use safe?
A: It can be effective, but carries risks. Systematic evaluation within clinical trials is crucial to monitor efficacy and toxicity.
Q: What is genomic profiling?
A: Analyzing a tumor’s DNA to identify genetic alterations that can be targeted with specific drugs.
Q: What is the DRUP trial?
A: A Dutch, pan-cancer clinical trial investigating the efficacy and safety of targeted therapies outside their registered indication.
Did you know? Approximately 39.1% of patients participating in the DRUP trial had rare cancers, a population that often benefits disproportionately from precision oncology approaches.
Pro Tip: If you’re considering off-label treatment, discuss the potential benefits and risks with your oncologist and explore whether you’re eligible for a clinical trial.
Want to learn more about precision oncology and clinical trials? Explore our other articles or subscribe to our newsletter for the latest updates.