The Future of Hope: Accelerating Gene Therapy for Children
Rare pediatric diseases, once considered medical mysteries, are increasingly becoming targets for groundbreaking cell and gene therapies. Recent advancements are dramatically shortening the time it takes to develop and deliver these potentially life-saving treatments, offering a beacon of hope for families facing previously untreatable conditions.
From Years to Months: The Speed of Innovation
The traditional drug development model struggles to accommodate the unique challenges of rare diseases, particularly in children. However, the landscape is shifting. A tailored gene therapy for an ultra-rare neurological disease was developed and administered to a patient within just three years. Even more remarkably, a patient-specific base-editing therapy for a lethal metabolic disorder was created, approved by regulators, and delivered to a newborn in approximately eight months. These examples demonstrate a significant acceleration in the development and delivery of personalized therapies.
The Commercial Viability Challenge
Despite these successes, biotech companies pioneering gene therapies often face financial hurdles. The individualized nature of these treatments clashes with the traditional for-profit drug development model, sometimes leading companies to withdraw products despite their proven efficacy. This highlights a critical market failure – the inability to sustainably fund the development and delivery of therapies for minor patient populations.
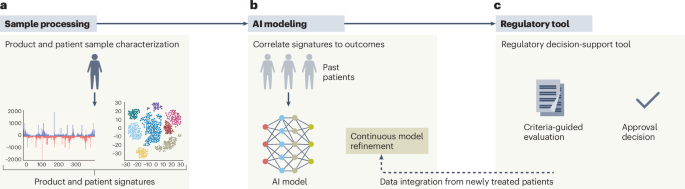
The UNICORN Framework: A New Approach
Researchers are proposing new frameworks to address these challenges. One such model, dubbed UNICORN, aims to streamline the process from product characterization to regulatory decision-making. This framework emphasizes a more efficient and collaborative approach to gene therapy development.
Pediatric Advanced Medicines Biotech: A Potential Solution
A proposed solution gaining traction is the creation of a new entity – the Pediatric Advanced Medicines Biotech (PAMB). This organization would focus on leading the late-stage development and commercialization of pediatric cell and gene therapies, operating outside the constraints of the traditional biopharmaceutical model. PAMB would partner with academic institutions, utilize academic good manufacturing practice facilities, and work closely with regulatory bodies to overcome the “valley of death” that often prevents promising therapies from reaching patients.
Licensing Models and Access
Current licensing practices often fail to adequately incorporate pediatric development milestones, further hindering progress. New licensing models are needed to incentivize investment in pediatric therapies and ensure equitable access for children in require. The FDA has already begun to recognize the urgency, awarding products Orphan Drug Designation, Rare Pediatric Disease Designation, and Breakthrough Therapy Designation based on promising clinical evidence.
Expanding Therapeutic Areas
Gene therapy is showing promise across a wide range of pediatric genetic disorders, including those affecting hematology, oncology, vision, hearing, immunodeficiencies, neurology, and metabolism. Ongoing clinical studies and approved drugs are continually expanding the possibilities for treatment and cure.
Did you know? The development of patient-specific therapies is becoming increasingly feasible, offering hope for children with ultra-rare conditions that were previously untreatable.
FAQ
Q: What is gene therapy?
A: Gene therapy involves introducing genetic material into cells to treat or prevent disease.
Q: Why are pediatric gene therapies particularly challenging?
A: Small patient populations and high development costs make it difficult for traditional pharmaceutical companies to invest in these therapies.
Q: What is the role of the FDA in accelerating these therapies?
A: The FDA is offering designations like Orphan Drug, Rare Pediatric Disease, and Breakthrough Therapy to incentivize development and expedite review.
Pro Tip: Stay informed about clinical trials and advocacy groups working to advance gene therapy for rare diseases. These organizations can provide valuable resources and support.
Q: What is the “valley of death” in drug development?
A: This refers to the stage where promising research fails to attract sufficient funding to progress to late-stage development and commercialization.
Want to learn more about the latest advancements in gene therapy? Explore more articles on Nature.com. Share your thoughts and experiences in the comments below!